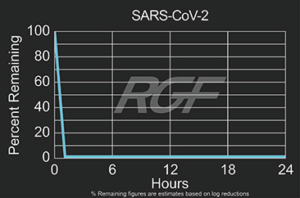
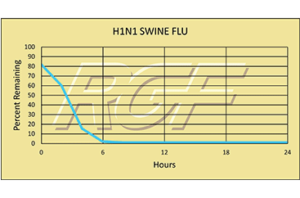
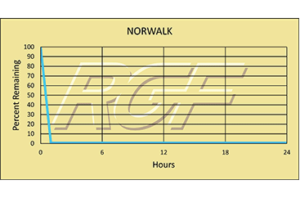
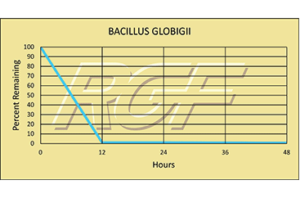
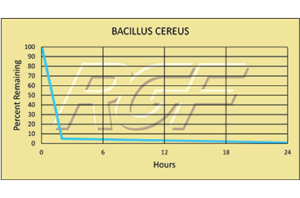
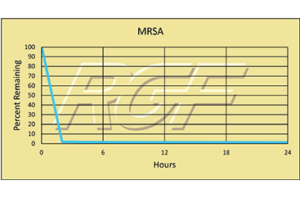
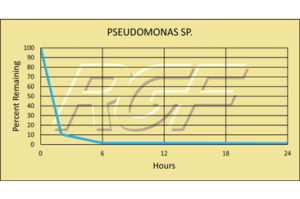
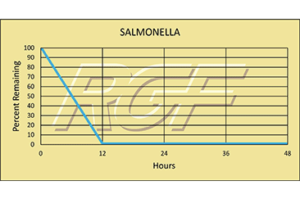
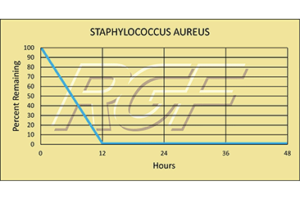
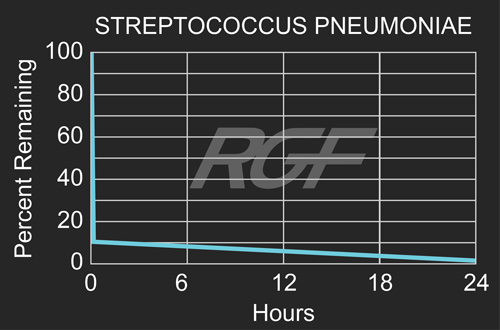
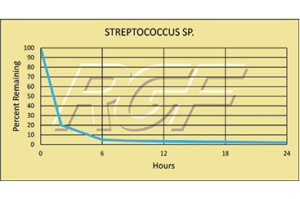
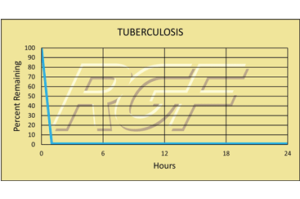
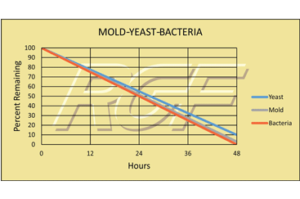
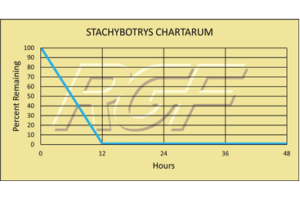
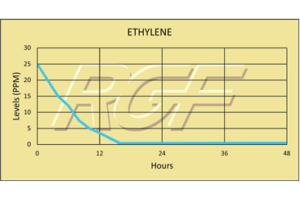
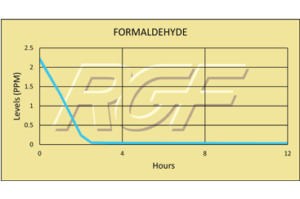
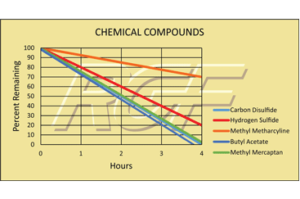
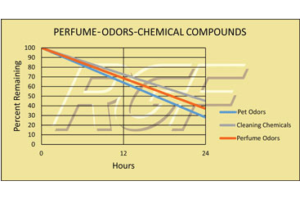
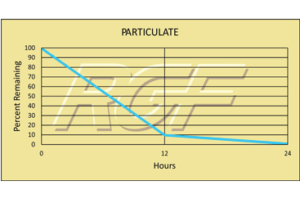
Testing Summary: Passed Federal Safety Standards
RGF Advanced Oxidation Technology devices have been thoroughly tested for ozone / emf – Electro Magnetic Frequency and have passed Federal Safety Standards
Tested by: ETL, TUV to UL standards, CSA, Kansas State University, and other third parties to ensure compliance with federal safety standards.
Note: Many household appliances emit some ozone and emf in safe low levels such as fluorescent lights, motors, computers, copy machines, refrigerators, blenders, electronic air filters, air conditioners, electric fans, microwave ovens, etc.